-
In January, a Parkville family learned their 8-year-old daughter had a rare illness. Though there is no cure, there is a drug that could help slow the disease. The problem? It costs more than $200,000 a year. Plus: Kansas politicians make bold claims about how to stop population declines in rural communities. Their efforts may not be enough to reverse the trend.
-
In January, the Anstaett family learned their eight-year-old daughter had Batten disease, a rare condition with a difficult — and eventually terminal — prognosis. The drug showing promise in slowing the disease isn't covered by their insurance because it's still in clinical trials.
-
The FDA banned donations entirely during the HIV epidemic in the early 1980s when little was known about AIDS. But the risk of transfusion-related transmission hasn’t been a real concern for decades. Kansas City University professor Dr. Benjamin Grin says the government’s holdout on changing guidelines is in part because of a lingering stigma.
-
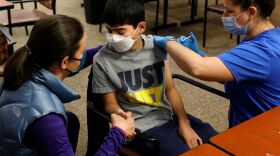
The FDA and CDC recently approved COVID-19 vaccines for children between the ages of six months to five years old. Vaccines are available through your pediatrician, some local pharmacies and hospitals.
-
The authorization comes in the midst of an explosion of COVID-19 cases nationwide driven by the omicron variant — a surge that has brought a spike in pediatric hospitalizations.
-
Not a single scientific or health authority in the U.S. recommends the use of the anti-parasitic drug ivermectin to treat or prevent COVID-19. Still, some Americans see the unproven drug as a way out of the pandemic.
-
The CDC is now recommending the Pfizer-BioNTech COVID-19 vaccine for the younger age group so we asked a pediatrician what should parents know before their kids get the shot.
-
With COVID-19 vaccines available soon for children ages 5-11, I wanted to see how Kansas City kids felt about their shots. I also heard something more: fears about the disease that's changed their lives, and impatience over long-promised plans and parties.
-
The agency acted after an independent panel of scientists strongly supported the move. Kids could start getting vaccinated within the week.
-
The Food and Drug Administration also gave an OK to boosters that differ from the vaccine originally used to immunize people against COVID-19. A mix-and-match approach could ease the booster rollout.
-
Pfizer and BioNTech say that early trial results show their vaccine established a strong antibody response against the coronavirus. FDA review is still needed.
-
Psychedelic drugs often have a negative connotation but ongoing clinical trials show they can be beneficial to some patients.
Play Live Radio
Next Up:
0:00
0:00
Available On Air Stations